ABOUT COMPANY
ABOUT COMPANY
From Research & Development and Production to Logistics, Marketing, Sales and International Distribution, Stago remains in control of its strategy at all levels.
Certified ISO 13485 and ISO 9001 (2008), the group’s industrial activites are mostly concentrated in France. Its geographical expansion has now led to opening R&D and production centers in the USA, Netherlands, Germany and Ireland. Its investment in R&D exceeds IVD sector’s standards. .
Since 1985, when the American affiliate was established, the direct distribution network has been expanded. In the last 10 years, 14 new affiliates have been established: China (2003), UK (2005), an office in Dubai for the Middle East (2007), Australia/New-Zealand and Canada (2008). Then an office in Hong Kong for Asia East (2011). In January 2012, Germany, Austria, Spain, Italy, Portugal, Switzerland, Belgium and Netherlands.
The companies belonging to the Stago Group are Stago, Tcoag, Agro-Bio, BioCytex, Thrombinoscope, Synapse and DSRV.
A Human Adventure
Founded by Jacques Viret at the end of the Second World War to market a solution to ease digestion and hepatic disorders, the Stago Group has now almost 2,200 employees, over half of whom are based in France.
The diversity of the men and women, professions and know-how is what allows Stago to develop, produce and sell the widest range of reagents and Haemostasis test instruments throughout the world using the most advanced technologies.
Customer satisfaction is a key value and everyone is conscious that there is a patient behind their actions.
Strength in Specialisation
With over 350 marketed products, Stago is a worldwide reference in Haemostasis and a 1stclass partner for biomedical laboratories.
Stago also has a licensed training center, offering theoretical and practical training courses at different levels.
Specialised in the fields of Haemostasis and Thrombosis, Stago invests in research and innovation to develop new and better performing reagents, systems and solutions. With 50 years of experience, Stago has acquired a charismatic image in Haemostasis and is well recognised among the international scientific world.
In this respect, Stago regularly organises symposiums or scientific meetings on Haemostasis research and latest practices, during conferences or as separate events.
Worldwide Presence
Stago is represented in over 110 countries via its affiliates and an extensive distribution network).
Each affiliate develops the processes implemented by that group, to provide our customers with the best support in terms of quality and services.
Each distributor is chosen on strict performance appraisal criteria with regards to their organization and staff: knowledge of Haemostasis, after-sales service capacities and commitment to promoting our products in a “customer satisfaction” culture. A specific internal structure (GSA) trains and monitors these teams.
-
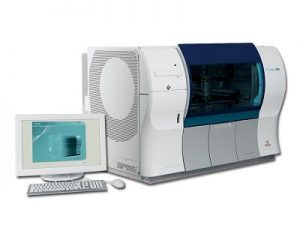
STA Compact Max3
STA Compact Max 3
Innovation born from Expertise
The STA Compact Max 3 offers significant performance improvements and includes the EPC (expert pre-analytical check) module.
This EPC module controls fill volumes of all types of tubes and detects haemolysed, icteric and lipemic samples, allowing the STA Compact Max 3 to check sample integrity to ensure quality results. It requires no additional plasma volume and does not affect throughput.
-
STA Satellite Max
STA Satellite Max | Efficiency born from Expertise
The Max family is now complete with the introduction of the STA Satellite Max. The STA Satellite Max builds on the proven strengths of the Max Generation, offering new functionalities.
The STA Satellite Max builds on the proven strengths of the Max Generation, offering new functionalities. and a user-friendly interface making it the ideal solution for Haemostasis testing in low-volume labs.
-
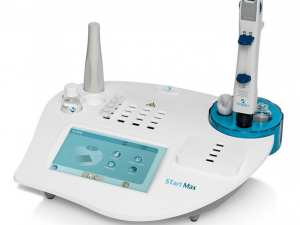
STart Max
STart Max
This small semi-automated instrument is just as impressive as the larger models: new quality control and calibration menus, and a large colour touchscreen to benefit from the standardised “Max Generation” graphical user interface & in terms of features nothing is missing either, with an effective traceability, the ability to archive patient results, quality controls, reagent lots, and a modern connectivity (to LIS and/or STA Coag Expert, with a USB port and the possibility to connect a handheld barcode reader).